Sedimentary Rocks
Sedimentary rocks are formed by the accumulation of sediments. There are three basic types of sedimentary rocks:1) Clastic sedimentary rocks such as breccia, conglomerate, sandstone and shale, that are formed from mechanical weathering debris;
2) Chemical sedimentary rocks such as rock salt and some limestones, that form when dissolved materials precipitate from solution; and,
3) Organic sedimentary rocks such as coal and some limestones which form from the accumulation of plant or animal debris.
Other (less abundant) type of sedimentary rocks are:
♦ Evaporite sedimentary rocks are composed of minerals formed from the evaporation of water. The most common evaporite minerals are carbonates, chlorides (halite and others built on Cl-), and sulfates (gypsum and others built on SO24-).
♦ Organic-rich sedimentary rocks have significant amounts of organic material, generally in excess of 3% total organic carbon. Common examples include coal, oil shale as well as source rocks for oil and natural gas.
♦ Siliceous sedimentary rocks are almost entirely composed of silica (SiO2), typically as chert, opal, chalcedony or other microcrystalline forms.
♦ Iron-rich sedimentary rocks are composed of >15% iron; the most common forms are banded iron formations and ironstones.
♦ Phosphatic sedimentary rocks are composed of phosphate minerals and contain more than 6.5% phosphorus; examples include deposits of phosphate nodules, bone beds, and phosphatic mudrocks
Clastic terrigenous sedimentary rocks consist of rock and mineral grains, or clasts, of varying size, ranging from clay-, silt-, and sand- up to pebble-, cobble-, and boulder-size materials. These clasts are transported by gravity, mudflows, running water, glaciers, and wind and eventually are deposited in various settings (e.g., in desert dunes, on alluvial fans, across continental shelves, and in river deltas).
Chemical sedimentary rock forms when mineral constituents in solution become supersaturated and inorganically precipitate. Common chemical sedimentary rocks include oolitic limestone and rocks composed of evaporite minerals such as halite (rock salt), sylvite, barite and gypsum.
Biochemical (Organic) sedimentary rocks are created when organisms use materials dissolved in air or water to build their tissue. Examples include:
• limestone formed from the calcareous skeletons of organisms such as corals, mollusks, and foraminifera.
• Coal which forms as plants remove carbon from the atmosphere and combine with other elements to build their tissue.
• Deposits of chert formed from the accumulation of siliceous skeletons from microscopic organisms such as radiolaria and diatoms.
Erosion ,Transport and deposition
Erosion is defined as the removal of soil, sediment, regolith, and rock fragments from the landscape. Erosion is responsible for the creation of hills and valleys. It removes sediments from areas that were once glaciated, shapes the shorelines of lakes and coastlines, and transports material downslope from elevated sites. Wind, water, and ice are the mediums primarily responsible for erosion. Finally, the process of erosion stops when the transported particles fall out of the transporting medium and settle on a surface. This process is called deposition.Transport: transport can occur in four different ways:
• Suspension is where the particles are carried by the medium without touching the surface of their origin. This can occur in air, water, and ice. Saltation is where the particle moves from the surface to the medium in quick continuous repeated cycles. The action of returning to the surface usually has enough force to cause the entrainment of new particles. This process is only active in air and water.
• Traction is the movement of particles by rolling, sliding, and shuffling along the eroded surface. This occurs in all erosional mediums.
• Solution is a transport mechanism that occurs only in aqueous environments. Solution involves the eroded material being dissolve and carried along in water as individual ions. Particle weight, size, shape, surface configuration, and medium type are the main factors that determine which of these processes operate.
Deposition: The erosional transport of material through the landscape is rarely continuous. Instead, we find that particles may undergo repeated cycles of transport and deposition. Transport depends on an appropriate balance of forces within the transporting medium. A reduction in the velocity of the medium, or an increase in the resistance of the particles may upset this balance and cause deposition. Reductions in competence can occur in a variety of ways.
Velocity can be reduced locally by the sheltering effect of large rocks, hills, stands of vegetation or other obstructions. Normally, competence changes occur because of large scale reductions in the velocity of flowing medium. For wind, reductions in velocity can be related to variations in spatial heating and cooling which create pressure gradients and wind. In water, lower velocities can be caused by reductions in discharge or a change in the grade of the stream. Glacial flows of ice can become slower if precipitation input is reduced or when the ice encounters melting.
Deposition can also be caused by particle precipitation and flocculation. Both of these processes are active only in water. Precipitation is a process where dissolved ions become solid because of changes in the temperature or chemistry of the water. Flocculation is a chemical process where salt causes the aggregation of minute clay particles into larger masses that are too heavy to remain suspended.
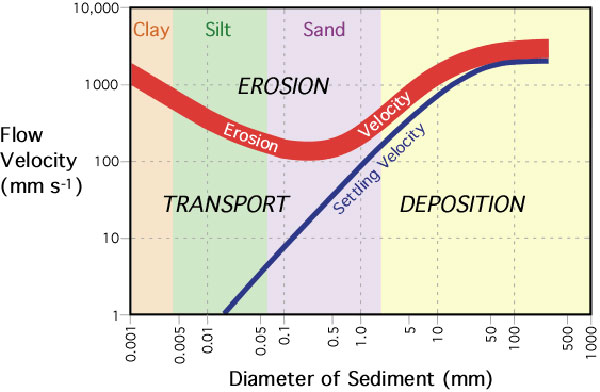
This graph describes the relationship between stream flow velocity and particle erosion, transport, and deposition. The curved line labeled "erosion velocity" describes the velocity required to entrain particles from the stream's bed and banks. The erosion velocity curve is drawn as a thick line because the erosion particles tends to be influenced by a variety of factors that changes from stream to stream. Also, note that the entrainment of silt and clay needs greater velocities then larger sand particles. This situation occurs because silt and clay have the ability to form cohesive bounds between particles. Because of the bonding, greater flow velocities are required to break the bonds and move these particles. The graph also indicates that the transport of particles requires lower flow velocities then erosion. This is especially true of silt and clay particles. Finally, the line labeled "settling velocity" shows at what velocity certain sized particles fall out of transport and are deposited.
Diagenesis: Sum of all processes, chiefly chemical, by which changes in a sediment are brought about after its deposition but before its final lithification (conversion to rock). Because most sediments contain mineral mixtures in which not all the minerals are in chemical equilibrium with each other, changes in interstitial water composition or changes in temperature or both will usually lead to chemical alteration of one or more of the minerals present. Diagenesis is considered a relatively low-pressure, low-temperature alteration process, whereas metamorphism is considered to be a rock-alteration process occurring at relatively higher pressures and temperatures. An example of diagenesis is the chemical alteration of a feldspar to form a distinctly new mineral in its place, a clay mineral.

